Gavin Outteridge, General Manager, Arysana
“Productivity is commonly defined as a ratio between the output volume and the volume of inputs. In other words, it measures how efficiently production inputs, such as labour and capital, are being used … to produce a given level of output.”i
My father worked on computers when there were 5 in the world. The US government had 4 and the British Ministry of Defence had the other one. If there was a problem with it, he picked up a screwdriver and walked into it to see what was going wrong. As computers became more widespread in the workplace in the 1970s, my mother (then a civil servant at the Ministry of Aviation) reported to me later that the general perspective on computer-aided productivity was that everyone would be working a 3-day week – getting the same output with less input.
Perhaps we’re greedier now – or embittered by the 3-day weeks we actually got in the late 70s when there wasn’t enough energy to keep offices open longer than that (good riddance to coal- and oil-fired power stations from the UK energy mix!) – but the theme of business today seems so often to be doing more with less. Is that possible with AI in drug development and commercialisation?
As Krugman neatly encapsulates it, there are two sides to it: inputs and outputs Inputs can be fewer and cheaper, outputs can be more and better and – in drug development with the patent clock ticking and the patient waiting – getting both sides faster is also a form of productivity gain.
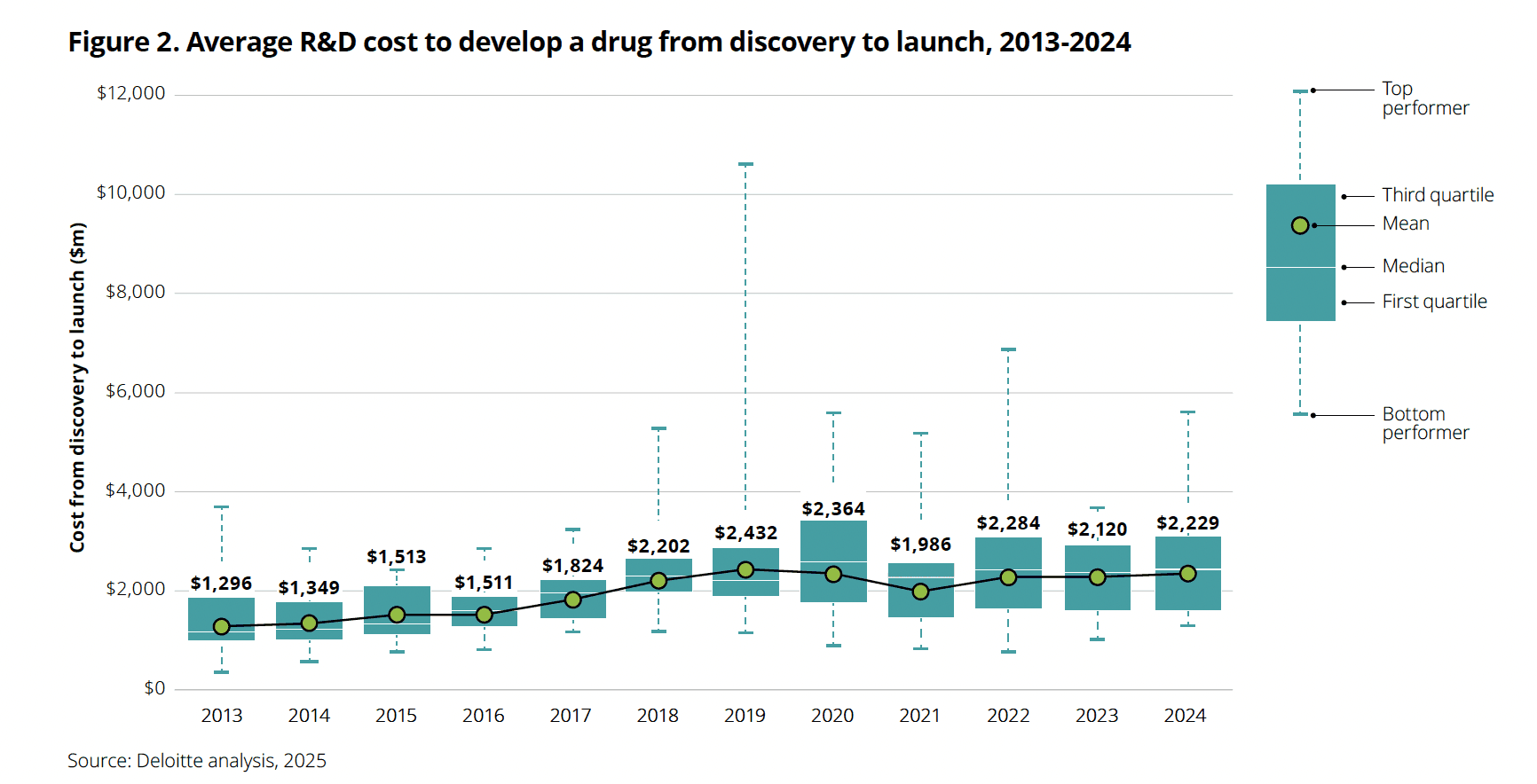
On the input side, it’s well-attested that compliant & safe drug development generally takes something like $2bn and around 10 years (if you think what happened during COVID could be the norm, that’s another conversation, focused more on myriad types of risk than on productivity; catch me during the networking drinks at our next conferenceii,iii and we can get into it!). Indeed, the 2025 edition of Deloitte’s annual ROI on Pharma R&D report states: “the average [drug development] cost per asset increased to $2.23 billion”iv during 2024-2025.
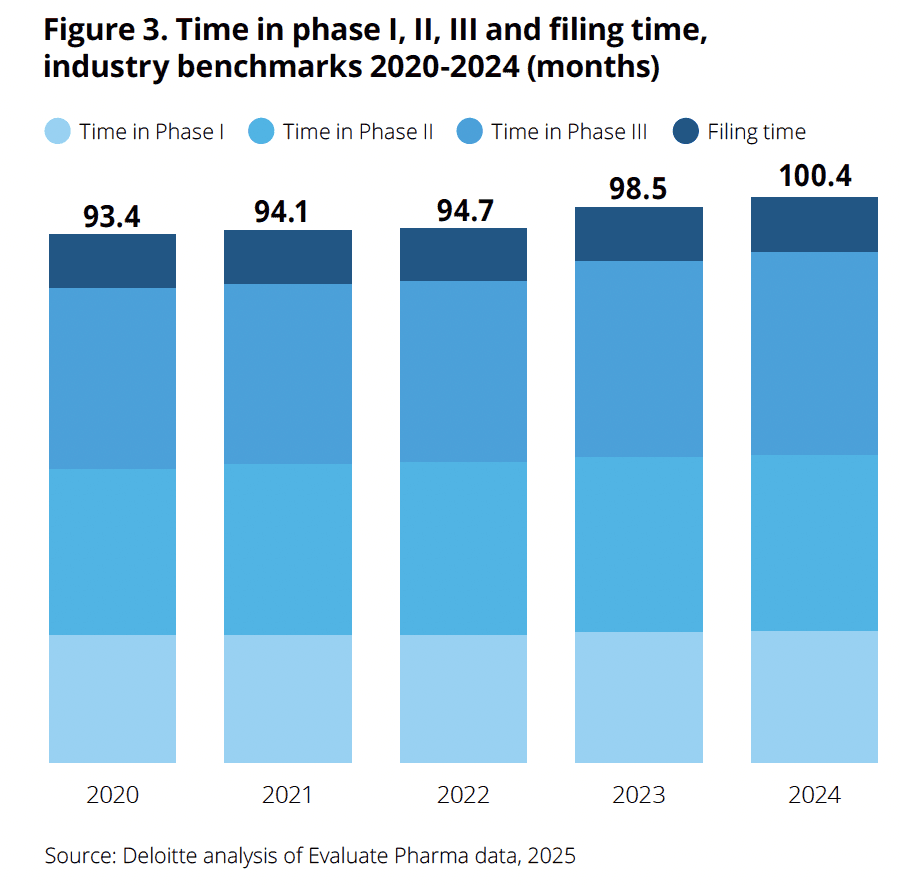
In the same report, Deloitte use Evaluate pharma data to show that “cycle times continue to increase year-on-year, and now exceed 100 months for time in phase I through to filing. While all stages of development have lengthened over the past five years, time in phase III has increased to the greatest extent, by 12.0 per cent”.v As any pharmaceutical executive will know, in drug development and commercialisation, time is definitely money – “a day later to market is a million dollars lost” I was told, during my onboarding at Pfizer in 2003.
So, for inputs to be usefully fewer, cheaper and faster, there’s apparently a lot of wriggle room but – to the compliant and safe piece – a whole load of things that need to be done and done very well.
But there is waste.
And there is inefficiency.
Strategists and scientists valued for their expertise and experience waste swathes of time making PowerPoint slides, updating Excel trackers or searching for documents in fragmented and labyrinthine systems; the same dataset gets purchased multiple times by the same company by different functions or different country teams; overlapping or entirely duplicative literature reviews get performed; three different teamsvi produce a set of messaging and evidence needs in static MS office documents, one called an IEGP, one called a Scientific Communications Platform (reader – it’s a slide deck, not a “platform!”) and one called a GVD. Outdated, disconnected and duplicative, time and time again, each piling up wasted dollars and – while you wait for results – letting time tick relentlessly away.
We’ve seen this in our old big pharma jobs, and in the global biotech and pharma companies we’ve consulted for and provide our tools to. Reducing this waste, and the attendant wasteful busywork, is a primary driver of the features and functions we’ve designed into our ATLAS platform.
Repeatable and scalable efficiency gains from things like:
Reduced $/hours spent on evidence search and summarisation, and related manual tasks
Reduced $ spend on duplicative or unnecessary evidence generation
Speed to availability of approved evidence and messaging from within the company, to those who need it outside the company: physicians, payers, patients, policymakers the general public… let’s call them “customers”!
Top-line productivity benefits
Identifying additional populations or increasing number of accessible markets by being better able to:
- Interrogate and communicate existing evidence
- Identify and then close gaps leading to increased access/uptake/reimbursed populationsDriving increased access, uptake and revenue by getting the right evidence to the right customer at the right time.
These are the productivity – bottom line AND top line – benefits that Arysana is targeting with the design of the ATLAS platform, and these same benefits are the kind of KPIs that our digital-forward biotech and pharma customers are using to prove positive impact.
So, is there addressable waste in pharma and biotech evidence generation? Yes.
Can it be tackled by deploying a digital platform for evidence and message management? Yes.
Is there an AI-enabled platform for integrated evidence generation planning that delivers measurable business benefits? Yes! Arysana’s ATLAS.
If you would like to join our community of biopharma innovators, and bend the curve of time and cost in drug development back towards greater productivity, we’d be delighted to hear from you!
Email me at gavin.outteridge@arysana.com or get in touch through our LinkedIn page.
ATLAS is an AI-enabled, integrated evidence planning software platform for pharmaceutical, biotech, diagnostics and medtech teams. Connect target product profiles, evidence generation tactics, scientific communication platforms and value messages in one system.
Designed to help executives and staff in Medical Affairs, Market Access, HEOR and IEP teams augment and accelerate their evidence-related workflows, ATLAS functions as an enhanced, dynamic, digital IEGP, SCP and GVD, reducing hours and dollars spent on message and evidence management, increasing strategic and cross-functional alignment and consistency, and – ultimately – increasing the ROI on evidence generation in pharmaceutical development.
References:
i Krugman, The Age of Diminishing Expectations (1994)
ii Reuters Pharma USA 2026
iii Faster, stronger value stories at scale: AI-enabled message & evidence alignment to augment & accelerate launch
iv Deloitte, Measuring the return from pharmaceutical innovation, 15th edition, March 2025
v Deloitte, ibid.
vi For more on the challenges of fragmented evidence generation planning, see Keyloun, ‘Why Does Integrated Evidence Planning for Pharma Still Feel Fragmented? Two Persistent Challenges’ January 2026


